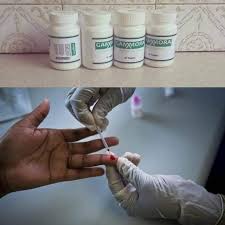
Public cautioned against taking ‘HIV cure’
Join our whatsapp news group for news on the go:
https://chat.whatsapp.com/FloefspSA811voJDItFGe1
The public has been warned by the Medicines Control Authority of Zimbabwe (MCAZ) against the use of Gamora, a drug developed by an Israel based company, allegedly to treat HIV.
The drug is being sold for US$4 150 in Harare from Sam Levy Village in Borrowdale and it is claimed that it can totally cure HIV within 21 days, with the user stopping to take Anti-retroviral treatment 14 days after taking the drug.
Gamora was developed by Zion Medical, an Israeli-based biotech company that recently claimed that the drug was tested on nine Ugandan patients at the Dr Ronald Bata Memorial Hospital in Entebbe.
An agent dealing with the drug said they did not register it with the MCAZ because it was not a locally manufactured drug.
“It has been confirmed by the world drug agency. Remember our treatment is taking 30 days to be cured. You are not allowed to take alcohol during the treatment period and keep your body healthy and strong by eating in time, we are 100 percent you will be permanently cured. Remember our medicine has three different types of cures, type A, B and C.
Type A it’s for those who are taking ARVs, type B is for those who’re yet to take ARVs or under three months on ARV treatment while type C is for people who are facing other types of strong desires like stroke,” said the agent to the Chronicle.
However, MCAZ spokesperson Mr Davison Kaiyo told Chronicle that police have since been engaged in line with its mandate of protecting public health.
Earlier, MCAZ issued a statement via its official Twitter account where it also encouraged members of the public to buy medicine from registered premises and persons.
“The mandate of the Medicines Control Authority of Zimbabwe (MCAZ) is to protect public health by ensuring that medicines and medical devices on the market are safe, effective and of good quality. Thus, all herbal medicines should be registered by the MCAZ. Gamora is currently not registered and not approved for sale in Zimbabwe.
“As MCAZ we have noted the continued advertising and marketing of such products, especially on social media. We urge members of the public to desist from buying such products which have not gone through any clinical trials and therefore their safety and efficacy has not been proven,” said the medicines control body.
It said the continued marketing and advertising of such unregistered medicinal products was not only an act of criminality but also puts the lives of citizens at risk.
“As the authority responsible for protecting public and animal health, MCAZ enforces adherence to standards by manufacturers and distributors and will exercise its statutory mandate against the perpetrators of these criminal acts,” said MCAZ.
According to official statistics, 1, 3 million people are HIV positive in Zimbabwe with 1, 2 million being on anti-retroviral treatment.
Zimbabwe is one of a handful of African countries that reached the UNAIDS’ 90-90-90 targets in 2021. It is also on track to meet the 2025 targets. The 90-90-90 targets aimed to bring HIV treatment to the vast majority of people living with HIV by the end of 2020 and to reduce the amount of HIV in their bodies to undetectable levels, so they keep healthy and prevent the further spread of the virus.
Zimbabwe’s 2025 targets are 95 percent of people with HIV will know their status, 95 percent of people with HIV who know their status will be on treatment and 95 percent of people with HIV on treatment will be virally suppressed.